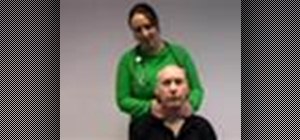
This video will teach and demonstrate the thyroid status assessment and thyroid gland examination in a patient. As a doctor, you will, at some point in your career, need to examine the thyroid gland in a patient's neck, and some other parts of the body which are effected by the thyroid.
In a general inspection, you'll be looking for visual clues, like whether the patient is properly dressed for the temperature, whether they're over or underweight, whether they appear anxious or agitated, if they have any facial myxedema, an odd complexion, and any hair loss (particularly the eyebrow). Then you'll check the hands, palms, wrist, and the neck area. Watch the video for the full thyroid examination procedure.
This video was produced by the Clinical Skills Online (CSO) project at the St. George's Hospital Medical School, in London, England.
Just updated your iPhone? You'll find new emoji, enhanced security, podcast transcripts, Apple Cash virtual numbers, and other useful features. There are even new additions hidden within Safari. Find out what's new and changed on your iPhone with the iOS 17.4 update.














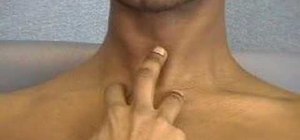





Be the First to Comment
Share Your Thoughts